What is Aluminum Nitride Ceramic?

Aluminum nitride (AIN) is known for its high thermal conductivity and remarkable electrical insulation properties. It is a common ceramic material, used in a variety of electrical devices. Aside from its thermal expansion coefficient and electrical insulation capabilities, aluminum nitride ceramics are resistant to attacks by most molten metals, such as copper, lithium, and aluminum. These, in combination with several other chemical and physical properties, make aluminum nitride a beneficial material in many industrial applications. In this article, you'll learn more about aluminum nitride, its different uses, properties, and specific use cases.
Overview of Aluminum Nitride (AlN)
Aluminum nitride (AlN) is a non-oxide ceramic material basically composed of aluminum and nitrogen (Al-65.81%, N-34.19), with the chemical formula AlN. It is a solid nitride of aluminum posing a high thermal conductivity of up to 321 W and also serves as an excellent electrical insulator. The aluminum nitride wurtzite phase has a bandgap of ~6 eV at room temperature. Because of these properties, this ceramic has proven useful in many applications, such as optoelectronics operating at deep ultraviolet frequencies.
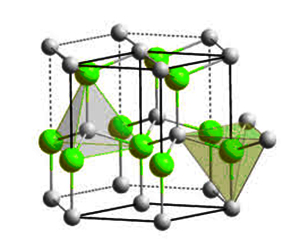
Aluminum nitride structure
Aluminum nitride was first synthesized by F. Briegleb and A. Geuther in 1862. The ceramic material exists mainly in the hexagonal wurtzite crystal structure. However, it is also available in the metastable cubic zincblende phase, which is mainly synthesized as thin films. Studies predict that the cubic phase of AlN can actually exhibit superconductivity at high pressures. The polycrystalline structure of aluminum nitride (AlN) has high thermal conductivity, accounting for its electrical insulating property. In terms of density, aluminum nitride is a covalent bond compound with a theoretical density of 3.26g/cm3.
Properties of Aluminum Nitride Ceramics
Aluminum nitride possesses several properties that make it suitable for a variety of industrial applications:
- High thermal conductivity (above 170W/m.K). This is close to the values for BeO and SiC and more than five times that of aluminum oxide (Al2O3).
- It has a thermal expansion coefficient of 4.5 *10-6℃, which is the same as that of Silicon (3.5-4 *10-6℃).
- It has good light transmission characteristics
- It does not possess toxic properties.
- Great electrical conductivity. The electrical properties of aluminum nitride include its dielectric constant, dielectric loss, bulk resistivity, and dielectric strength - all of which make it an excellent insulating material.
- Good mechanical properties: The mechanical properties of aluminum also account for its wide use in industrial processes. It has a higher bending strength than aluminum oxide (Al2O3) and beryllium oxide (BeO) ceramics.
Why Aluminum Nitride Ceramics are Widely Used
Aluminum nitride ceramics have been used in many fields due to their favorable properties, including high thermal conductivity, low dielectric constant and dielectric loss, high insulation strength, and their remarkable resistance to plasma erosion. Some of the common applications of this material are:
- chipping heat dissipation and support
- the use of aluminum nitride ceramic substrates (ceramic trays) in semiconductor devices
- aluminum nitride etching shields
- aluminum nitride evaporation boats for OLEDs
Another vital use case for aluminum nitride ceramics is in the manufacture of plastics and resins. These materials typically have low thermal conductivity (less than 0.3W/mk). By adding aluminum nitride powder to plastics and resins, they produce thermally conductive potting compounds and thermal pads having high thermal conductivity, which is typically above 10W/mk. They are also used in the packaging of various electronic components. More researchers are examining the production of light-emitting diodes to operate in the ultraviolet region using gallium nitride-based semiconductors. These studies show that it might be possible to achieve wavelengths as short as 250 nm using the aluminum gallium nitride alloy.
Further Applications of Aluminum Nitride Ceramics
Aluminum nitride has been used extensively in the following applications:
- Heat sinks and heat spreaders
- Manufacturing of steel and semiconductor
- Electrical insulators
- Silicon wafer handling and processing
- Used as crucibles for growing crystals of gallium arsenide
- Serves as substrates and insulators for microelectronic devices
- Substrates for electronic packages
- Chip carriers for sensors and detectors
- Laser heat management components
- Functions as dielectric layers in optical storage media
- Molten metal fixtures
- Packaging for microwave devices
- Opto-electronics

Aluminum nitride crucible
Aluminum Nitride Ceramic Component Machining
Depending on the method of production and the desired properties, the properties of aluminum nitride can be seen in three different forms:
- Conventional aluminum nitride ceramics are produced by casting and dry pressing.
- Injection-molded aluminum nitride ceramics effectively solve the problem of high hardness, brittleness, and complex processing of aluminum nitride ceramics.
- Injection molding high thermal conductivity aluminum nitride ceramics is based on the injection molding aluminum nitride ceramic process and uses high-purity, higher-quality aluminum nitride powder. The product has varying properties that depend on the product configuration and manufacturing process.
Today, many manufacturers have mastered producing technical ceramics and constantly provide precision ceramic products to several companies. Organizations or manufacturers that need aluminum nitride ceramic machining, technical ceramic prototyping, ceramic casting, and ceramic metal packaging can turn to trusted ceramics manufacturers for quality technical aluminum ceramic materials.
Conclusion
The high thermal conductivity, high thermal resistance, low expansion coefficient, high mechanical strength, resistance to chemical corrosion, and low dielectric loss of aluminum nitride ceramics make them an ideal choice in various industrial applications. Thank you for reading our article and we hope it can help you to have a better understanding of aluminum nitride ceramics. For more information, please visit https://www.preciseceramic.com/.
{{item.content}}
LEVE A REPLY
{{item.children[0].content}}
{{item.content}}
LEAVE A REPLY
SUBSCRIBE OUR NEWSLETTER
- How PBN Crucibles Ensure the Quality of GaN & SiC Epitaxial Materials
- SiC vs. Quartz Focus Rings: A Cost and Performance Analysis for Advanced Etch
- AlN Ceramic Substrates: Enabling Next-Gen Electrostatic Chucks
- The Amor of Semiconductor Tools: Why High-Purity Al2O3 & AlN Are Preferred for Plasma Process Chambers
- Silicon Carbide - Ultra-High Temperature Ceramics for Extreme Environments