Types and Applications of All Kinds of Ceramic Materials
Ceramics greatly differ in their basic composition, and their properties also vary greatly due to differences in bonding. Classification of ceramics based on their specific applications and composition is one of the two most important ways among many ceramic classification methods.
Classification of Ceramics by Composition
Based on their composition, ceramics are classified as silicates, oxides, carbides, nitrides, borides, etc

Silicate Ceramics
Silicates are materials generally having a composition of silicon and oxygen. The main types of silicate ceramics are based either on alumosilicates or magnesium silicates. They are traditionally categorized into coarse or fine and, according to water absorption, into dense (<2% for fine and <6% for coarse) or porous ceramics (>2% and >6%, respectively).
Further Reading: What are Silicate Ceramics
Oxide Ceramics
Oxide ceramics include alumina, zirconia, silica, magnesia, and other metal oxide-based materials. These are non-metallic, inorganic compounds in which oxygen is the primary anion, combined with metallic or semimetallic elements.
Oxide ceramics possess the following properties:
- (a) High melting points
- (b) High wear resistance (low wear rate)
- (c) An extensive collection of electrical properties
These types of ceramics are available with a variety of special features. For example, glazes and protective coatings seal porosity, improve water or chemical resistance, and enhance joining to metals or other materials. Oxide ceramics are used in a wide range of applications, which include materials and chemical processing, radiofrequency and microwave applications, electrical and high voltage power applications, and foundry and metal processing. Aluminum oxide (Al2O3) is the most important technical oxide ceramic material. This synthetically manufactured material consists of aluminum oxide ranging from 80 % to more than 99 %.
Non-Oxide Ceramics
Non-oxide ceramics include materials such as carbides, nitrides, and borides. The use of non-oxide ceramics has enabled extreme wear and corrosion problems to be overcome, even at high temperatures and severe thermal shock conditions. These types of ceramics find their application in different spheres such as pharmaceuticals, the oil and gas industry, valves, seals, rotating parts, wear plates, location pins for projection welding, cutting tools, abrasive powder blast nozzles, metal forming tooling, etc.
Glass-ceramics
These are polycrystalline materials manufactured through the controlled crystallization of base glass. Glass-ceramic materials share many common characteristics with both glass and ceramics. Glass-ceramics possess an amorphous phase and more than one crystalline phase. These are produced by a controlled crystallization procedure. Glass-ceramics hold the processing advantage of glass and have special characteristics of ceramics.
For example, lithium aluminosilicate glass-ceramics are nucleated by TiO₂ or ZrO₂ and heat-treated to form fine crystals, yielding near-zero thermal expansion.
Classification of Ceramics by Applications
While the previous section categorized ceramics by their chemical makeup, a complementary approach is to classify them by their practical applications, as shown below.
The other important classification of ceramics is based on their application, such as glasses, clay products, refractories, abrasives, cement, and advanced ceramics.
In general, ceramic materials used for engineering applications can be divided into two groups: traditional ceramics and advanced ceramics. Typically, traditional ceramics are made from three basic components: clay, silica (flint), and feldspar. For example, bricks, tiles, and porcelain articles. However, advanced ceramic materials consist of highly pure compounds of aluminum oxide (Al2O3), silicon carbide (SiC), and silicon nitride (Si3N4).

Glasses
Glasses are a familiar group of ceramics – containers, windows, mirrors, lenses, etc. They are non-crystalline silicates containing other oxides, usually CaO, Na2O, K2O, and Al2O3, which influence the glass properties and its color. A typical property of glasses that is important in engineering applications is their response to heating. There is no definite temperature at which the liquid transforms into a solid, as with crystalline materials. A specific temperature, known as the glass transition temperature (Tg), is defined based on viscosity above which the material is named as supercooled liquid or liquid, and below it is termed as glass.

Clay Products
Clay is one of the most widely used ceramic raw materials. It is found in great abundance and is popular because of the ease with which products are made. Clay products are mainly of two kinds: structural products (bricks, tiles, sewer pipes) and whitewares (porcelain, chinaware, pottery, etc.).

Refractories
These are described by their capacity to withstand high temperatures without melting or decomposing, and their inertness in severe environments. Thermal insulation is also an important function of refractories.
Abrasive Ceramics
Abrasive ceramics are used to grind, wear, or cut away other materials. Thus, the prime requisite for this group of materials is hardness or wear resistance in addition to high toughness. As they may also be exposed to high temperatures, they need to exhibit some refractoriness. Diamond, silicon carbide, tungsten carbide, silica sand, and aluminum oxide/corundum are some typical examples of abrasive ceramic materials.
Cement
Cement, plaster of paris, and lime come under this group of ceramics. The characteristic property of these materials is that when they are mixed with water, they form a slurry that sets subsequently and hardens finally. Thus, it is possible to form virtually any shape. They are also used as bonding phases, for example, between construction bricks.

Advanced Ceramics
Advanced ceramic materials are newly developed and manufactured in a limited range for specific applications. Usually, their electrical, magnetic, and optical properties, and a combination of properties, are exploited. Typical applications: heat engines, ceramic armors, electronic packaging, etc.
Further Reading: An Overview of Advanced Ceramic Materials
Examples of Specific Ceramics and Respective Applications
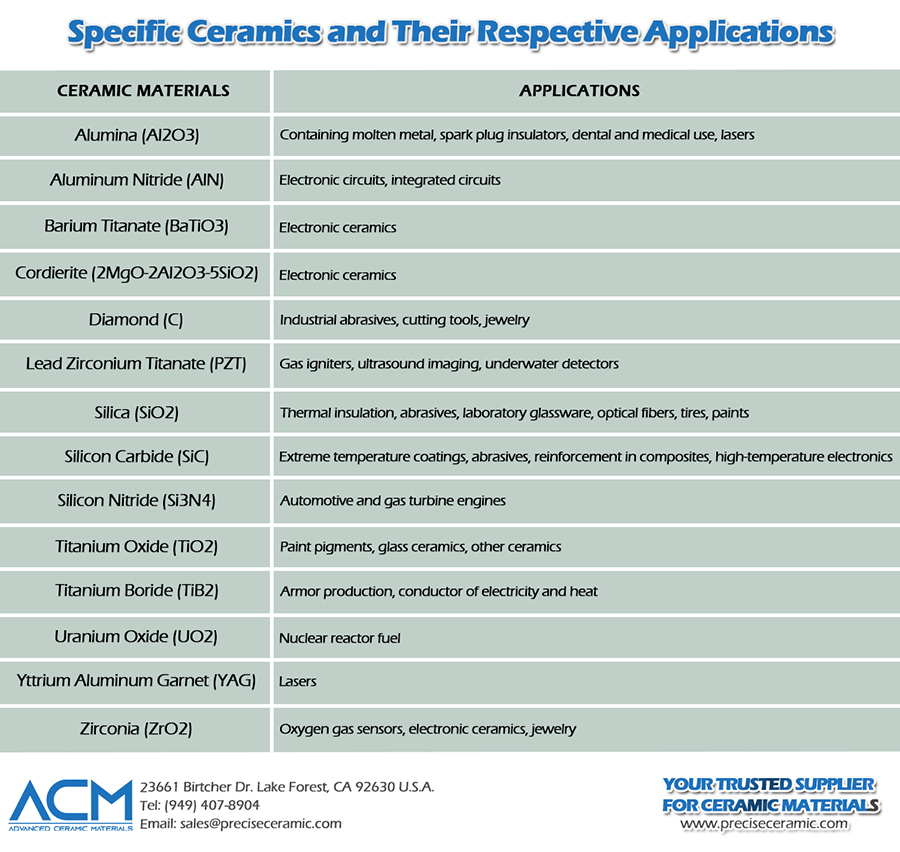
Aluminum oxide / Alumina (Al2O3)
Alumina is one of the most commonly used ceramic materials. It is used in many applications, such as to contain molten metal, where the material is operated at very high temperatures under heavy loads, as insulators in spark plugs, and in some unique applications such as dental and medical use. Chromium-doped alumina is used for making lasers. Typical commercial grades range from 85% to 99.9% purity, with a Vickers hardness of ~15–19 GPa and a maximum service temperature of ~1700°C.
Aluminum nitride (AlN)
Because of its typical properties, such as good electrical insulation but high thermal conductivity, aluminum nitride is used in many electronic applications, such as in electrical circuits operating at high frequency. It is also suitable for integrated circuits. Other electronic ceramics include barium titanate (BaTiO3) and Cordierite (2MgO-2Al2O3-5SiO2). Aluminum nitride offers a thermal conductivity of 140–180 W/m·K and electrical resistivity >10¹⁴ Ω·cm.
Diamond (C)
It is the hardest material known to be available in nature. It has many applications, such as industrial abrasives, cutting tools, abrasion-resistant coatings, etc. It is, of course, also used in jewelry.
Lead Zirconium Titanate (PZT)
PZT is the most widely used piezoelectric material and is used as a gas igniter, for ultrasound imaging, and in underwater detectors.
Further Reading: Piezoelectric Materials and Their Diverse Applications
Silica (SiO2)
Silicon dioxide is an essential ingredient in many engineering ceramics and thus is the most widely used ceramic material. Silica-based materials are used in thermal insulation, abrasives, laboratory glassware, etc. It also found application in communications media as an integral part of optical fibers. Fine particles of silica are used in tires, paints, etc.
Silicon Carbide (SiC)
Silicon carbide is known as one of the best ceramic materials for very high-temperature applications. It is used as a coating on other materials for protection from extreme temperatures. It is also used as an abrasive material. It is used as a reinforcement in many metallic and ceramic-based composites. It is a semiconductor and is often used in high-temperature electronics. Silicon nitride (Si3N4) has properties similar to those of SiC but is somewhat lower, and is found in applications such as automotive and gas turbine engines. It has a thermal conductivity of 120–270 W/m·K and can be used up to ~1600°C in air.
Titanium Oxide (TiO2)
Titanium dioxide is mostly found as a pigment in paints. It also forms part of certain glass-ceramics. It is used to make other ceramics like BaTiO3.
Titanium Boride (TiB2)
It exhibits great toughness properties and, hence, is found in applications in armor production. It is also a good conductor of both electricity and heat.
Uranium Oxide (UO2)
It is mainly used as nuclear reactor fuel. It has exceptional dimensional stability because its crystal structure can accommodate the products of the fission process.
Yttrium Aluminum Garnet (YAG, Y3Al5O12)
Yttrium Aluminum Garnet has a main application in lasers (Nd-YAG lasers).
Zirconia (ZrO2)
It is also used in producing many other ceramic materials. Zirconia is also used in making oxygen gas sensors, as an additive in many electronic ceramics. Its single crystals are part of the jewelry. Zirconia exhibits high fracture toughness (7–12 MPa·m¹/²), significantly higher than that of alumina.
Conclusion
To conclude, ceramics are a diverse group of materials with an extensive range of physical and chemical properties that make them useful in many different applications. The classification of ceramics based on their composition and application provides a better understanding of the characteristics and potential uses of each type. From glasses for containers and lenses to advanced ceramics for heat engines and electronic packaging, ceramics are ubiquitous in our daily lives, and ongoing research and development continue to expand their potential applications.
{{item.content}}
LEVE A REPLY
{{item.children[0].content}}
{{item.content}}
LEAVE A REPLY
SUBSCRIBE OUR NEWSLETTER
- Case Study: Sub-Millimeter Sapphire Tubes for High Voltage Discharge Applications
- Alumina Powder (Al2O3): A Technical Guide to Grades, Properties, and Applications
- Ceramic Machining: Essential Techniques and Processes for Precise Components
- The Melting Behavior of Ceramics: A Technical Guide for High-Temperature Applications
- AlN Ceramic Substrates for UV-LED and Mini LED Packaging