An Overview of Silicon Carbide Ceramic Materials
What is Silicon Carbide Ceramic?
Silicon carbide ceramic (SiC) is an advanced ceramic material containing silicon and carbon. It occurs in nature as the extremely rare mineral moissanite. Synthetic SiC powder has been mass-produced since 1893 for use as an abrasive. Grains of silicon carbide can be bonded together by sintering to form very hard ceramics.
With the rapid development of modern space technology, the automobile industry, and marine engineering, the requirements for materials are increasing. It is urgent to develop all kinds of new high-performance structural materials.
Silicon carbide ceramic materials have many excellent features, such as high-temperature strength, good wear resistance, small thermal expansion coefficient, high hardness, thermal shock, chemical corrosion resistance, etc. Therefore, it is widely used in the automobile, mechanica,l and chemical industries, environmental protection, space technology, information electronics, energy, and other fields. Silicon carbide has become an irreplaceable structural ceramic with excellent performance in many industrial fields.
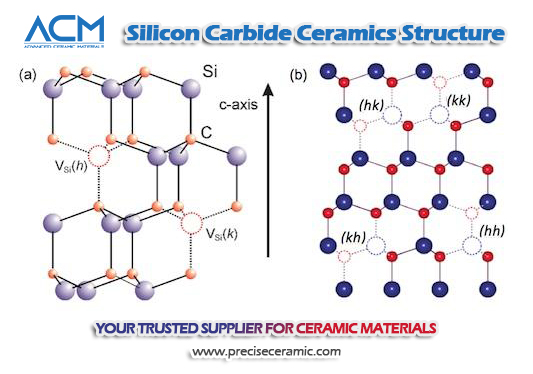
For specific examples, silicon carbide ceramics have been widely used as corrosion-resistant containers and pipelines in the petrochemical industry, and have also been successfully used in various bearings, cutting tools, and mechanical seal parts in the mechanical industry. Besides that, silicon carbide is also considered to be the most promising candidate for the future production of gas turbines, rocket nozzles, and engine components in the aerospace and automotive industries.
Basic Properties of Silicon Carbide Ceramics
Chemical properties
When the temperature in the oxygen reaction reaches 1300 ℃, a silicon dioxide protective layer will be formed on the surface of the silicon carbide crystal. With the thickening of the protective layer, the silicon carbide inside resisted continuing to be combined, which makes the silicon carbide have good anti-chemical property. In terms of acid, alkali, and compound resistance, silicon carbide has strong acid resistance but poor alkaline resistance because of the utility of silica protective film.
Physical properties
The density of various silicon carbide crystals is similar, which is usually 3.20 g/mm. Silicon carbide has a hardness of 9.5 Mohs, and the hardness of Knoop is 2670 - 2815 kg/mm, which is higher than that of corundum in abrasive materials, and second only to diamond, cubic boron nitride, and boron carbide. The thermal conductivity and thermal shock resistance of silicon carbide ceramic are very high, and the thermal expansion parameters are small, so SiC ceramic is a high-quality refractory material.
Electrical properties
Industrial silicon carbide ceramic at constant temperature is a kind of semiconductor, which belongs to impurity conductivity. The internal resistance of high-purity silicon carbide decreases with the increase in temperature. The conductivity of silicon carbide with different impurities is also different.
Good hydrophilicity
As is known to all, SiC is a compound with strong covalent bonds. According to Pauling's calculation of electronegativity, the ionic property of the Si-C bond in SiC is only about 12%. Therefore, SiC has high hardness, a large elastic modulus, and good wear resistance. It is worth pointing out that the silicon dioxide layer formed on the surface will inhibit the further diffusion of oxygen when SiC is oxidized, so the oxidation rate is low.
Developing History of Silicon Carbide Ceramics
Silicon carbide is a kind of carbide accidentally discovered by American Acheson in the 1891 fused diamond experiment. At that time, it was mistaken for a mixture of diamonds. In 1893, Acheson developed a method for industrial smelting of silicon carbide, which is commonly known as the Acheson furnace.
This method has been used until now. It uses a carbonaceous material as the core of the resistance furnace and electrically heats the mixture of quartz, SiO2, and carbon to form silicon carbide.
Several important events about silicon carbide:
1905 The first time silicon carbide was found in meteorites.
1907 The first silicon carbide crystal light-emitting diode was born.
1955 A major breakthrough in theory and technology, LELY proposed the concept of high-quality carbonization, and since then SiC has been used as an important electronic material.
1958 The first World Silicon Carbide Conference was held in Boston for academic exchanges.
1978 In the 1960s and 1970s, silicon carbide was mainly studied by the former Soviet Union. By 1978, the grain purification method of "LELY improvement technology" was first adopted. From 1987 to the present, the silicon carbide production line was established with the research results of CREE, and suppliers began to provide commercial silicon carbide substrates.
Application Fields of Silicon Carbide Ceramic
There are four main application areas for silicon carbide ceramic: functional ceramics, advanced refractory materials, abrasives, and metallurgical raw materials. The raw material of silicon carbide can be supplied in large quantities, and it cannot be regarded as a high-tech product, and the application of nano-sized silicon carbide powder with extremely high technical content cannot form economies of scale in a short time.
![]()
(1) As an abrasive, it can be used as a grinding tool, such as a grinding wheel, oil stone, grinding head, sand tile, etc.
(2) As a metallurgical deoxidizer and high-temperature resistant material.
(3) A high-purity single crystal that can be used for manufacturing semiconductors and silicon carbide fibers.
Main uses: for 3-12 inch single crystal silicon, polysilicon, potassium arsenide, quartz crystal and other wire cutting. Solar photovoltaic industry, semiconductor industry, piezoelectric crystal industry engineering processing materials. Used in semiconductors, lightning rods, circuit components, high-temperature applications, UV detectors, structural materials, astronomy, disc brakes, clutches, diesel particulate filters, filament pyrometers, ceramic membranes, cutting tools, heating elements, nuclear fuel, jewelry, steel, protective gear, catalyst carriers, and other fields.
Conclusion
Thanks for reading our articles, and we hope that you now know more about silicon carbide ceramic after reading this article. If you want to know more about SiC ceramic or other advanced ceramic materials, we would like to recommend that you visit http://www.preciseceramic.com for more information.
{{item.content}}
LEVE A REPLY
{{item.children[0].content}}
{{item.content}}
LEAVE A REPLY
SUBSCRIBE OUR NEWSLETTER
- How PBN Crucibles Ensure the Quality of GaN & SiC Epitaxial Materials
- SiC vs. Quartz Focus Rings: A Cost and Performance Analysis for Advanced Etch
- AlN Ceramic Substrates: Enabling Next-Gen Electrostatic Chucks
- The Amor of Semiconductor Tools: Why High-Purity Al2O3 & AlN Are Preferred for Plasma Process Chambers
- Silicon Carbide - Ultra-High Temperature Ceramics for Extreme Environments