Do you have any questions? Email Us
[email protected]

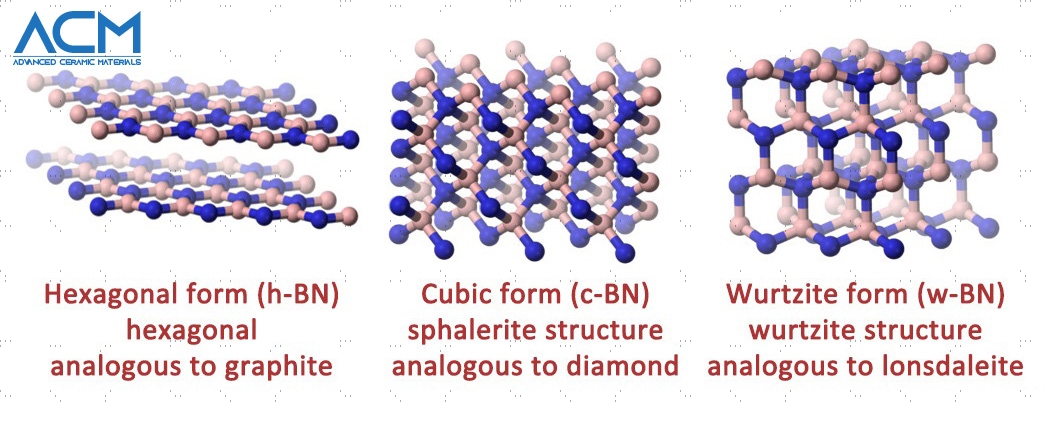
Pyrolytic boron nitride is abbreviated as Pyrolytic BN or PBN, and also known as Chemical vapour-deposited Boron Nitride, Chemical Vapour-deposition of Boron Nitride or CVD-BN. As the name suggests, this is a kind of boron nitride prepared by high temperature pyrolysis reaction by chemical vapor deposition. Speaking of boron nitride, we must first talk about the two main structures of boron nitride: hexagonal and cubic. This is very similar to the two crystal forms of carbon: graphite and diamond. Hexagonal boron nitride and graphite are very similar in many respects, and hexagonal boron nitride is therefore named white graphite. The pyrolytic boron nitride is hexagonal boron nitride, but strictly speaking, it has a certain amount of disordered layer structure. The main feature of PBN prepared by chemical vapor deposition is that the purity is very high and can reach more than 99.99%.
Pyrolytic boron nitride (PBN) belongs to the hexagonal system and is a typical layered material. The inter-layer and inter-layer atomic spacing is quite different, the inter-layer atom spacing is 3.33, and the intra-layer atom spacing is 1.45. The structure of the PBN consists of alternating B and N atoms in the layer and along the C axis, respectively, and the stacking method is ababab. It can be observed under a high power microscope that PBN exhibits stacking faults, resulting in an average layer spacing greater than the theoretical layer spacing. Since the PBN grows in the C-axis direction during the deposition process, it exhibits a high degree of layer orientation, and the preferred orientation direction is the C-axis.
PBN is an advanced material with excellent properties. It is deposited by the reaction of ammonia and boron halide under high temperature and high vacuum conditions. It can be deposited as PBN sheet material or directly into PBN final products such as tubes, rings or thin-walled containers. Unlike ordinary hot-pressed boron nitride (BN), it does not have to undergo a conventional hot-press sintering process without adding any sintering agent, so the obtained product has the following remarkable features:
(1) Non-toxic and odorless;
(2) High purity, reaching 99.999% or more;
(3) It does not react with acid, alkali, salt and organic reagents at room temperature, and is slightly corroded in molten salt or alkali solution, but it can resist corrosion of various acids at high temperature;
(4) does not react with most molten metals, semiconductors and their compounds;
(5) The oxidation resistance is good below 1000 °C;
(6) Good thermal shock resistance, no cracks were found in the water at 2000 °C;
(7) The use temperature is high, there is no sublimation point, and it is directly decomposed into B and N above 3000 °C;
(8) High resistance and good electrical insulation performance;
(9) Smooth surface, no pores, and not wet with most semiconductor melts.
Pyrolytic boron nitride (PBN) materials have high purity, chemical inertness and excellent structure and properties, making pyrolytic boron nitride crucible an ideal containers for elemental purification, compound and compound semiconductor crystal growth, especially for the preparation of next-generation compound semiconductors such as GaAs, InP, etc. At the same time, it is also widely used in aviation, aerospace, electronics, chemical, special metallurgy, medical and other fields. The main uses of PBN are as follows:
(1) Compound semiconductor single crystal growth container The growth of compound semiconductor single crystals (such as GaAs, InP, etc.) requires an extremely strict environment including temperature, raw material purity, and growth vessel purity. PBN crucible is the most ideal container for the growth of compound semiconductor single crystals, and it is irreplaceable. Compound semiconductor single crystal growth methods currently mainly include liquid-sealed straight pull method (LEC), horizontal Breman method (HB) and vertical Breman method (VB and VGF), and the corresponding PBN has LEC crucible, VB crucible and VGFc crucible and so on;
(2) Crucibles for molecular beam epitaxy Molecular beam epitaxy (MBE) is one of the most important epitaxial growth processes for III-V and II-VI semiconductor crystals in the world today. PBN crucible is the best container for the evaporation of elements and compounds in the MBE process;
(3) Crucibles for OLED Pyrolytic boron nitride crucible is used in OLED production lines as an evaporation element container.
(4) PBN ring and PBN sheet PBN can be heated to 2300 ° C under ultra-high vacuum without decomposition, has a high purity, more than 99.99%, does not release gas impurities and other excellent characteristics at high temperatures, these characteristics make PBN can be processed into different shapes. PBN rings and PBN sheets are widely used in high temperature, vacuum, MBE and OLED fields as support frames, insulators and gaskets.
(5) PBN coating In liquid phase epitaxy and MOCVD, PBN is commonly used to protect the graphite parts in the equipment, and can also be used as an outer coating of graphite heaters (PG/PBN heaters) to prevent the graphite heating elements from volatilizing at high temperatures. . In addition, since the PBN film has extremely high chemical inertness, it can be used as a separator or a passivation film in a semiconductor etching process; For more information, please visit https://www.preciseceramic.com/.